
Federal Exclusive: In COVID times, sanitiser makers work without licences
The Federal conducted an experiment. After we got a tipoff from dealers in the sanitisation industry that cases of adulteration have increased, we bought random samples of sanitisers from the market and gave it for testing to know if the manufacturers adhered to norms and maintained adequate alcohol content in their products.

‘Sanitise your hands’ is an often issued advisory amidst the COVID-19 mayhem with the coronavirus causing havoc globally. State government campaigns like ‘Break the Chain’ and ‘Wash hands, live long’ are aimed at spreading awareness about maintaining good sanitisation practices.
However, for the people to be safe, it is also important for the hand wash or sanitiser to be of standard quality.
So, The Federal conducted an experiment. After we got a tipoff from dealers in the sanitisation industry that cases of adulteration have increased, we bought random samples of sanitisers from the market and gave it for testing to know if the manufacturers adhered to norms and maintained adequate alcohol content in their products.
We bought these samples from different markets in Chennai and got them tested at a certified laboratory to test the percentage of alcohol content in them.

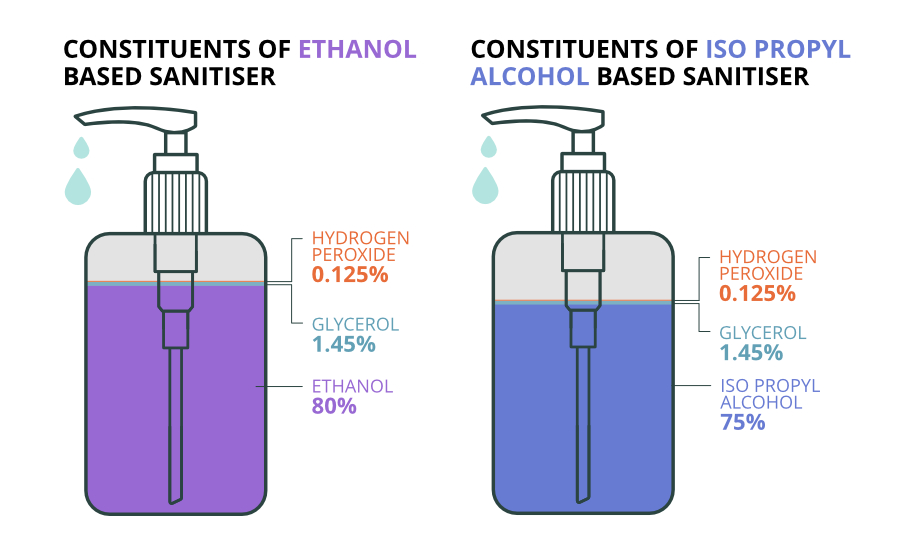
Constituents of a hand sanitiser
Alcohol is the key ingredient in a sanitiser that helps kill microbes. It also helps determine if the product is effective. Both Ethanol and Isopropyl alcohol are used to make sanitisers.
Apart from alcohol, sanitisers also contain glycerol for moisturising hands and hydrogen peroxide to kill germs. Only if a sanitiser has a mix of all these three ingredients in the right proportion, the sanitiser will be effective.
The World Health Organisation (WHO) has recommended two standards to make sanitiser solutions.
Ethanol 80% (v/v), Glycerol 1.45% (v/v) and Hydrogen peroxide 0.125% (v/v) or Isopropyl alcohol 75% (v/v), Glycerol 1.45% (v/v) and Hydrogen peroxide 0.125% (v/v)

As per the central drug regulator, DCGI, both formulations were first approved for sale in India in 2017. These formulations are preferred by most manufacturers and marketers due to the WHO endorsement.
Out of the six samples we tested, three did not adhere to the WHO recommendations.
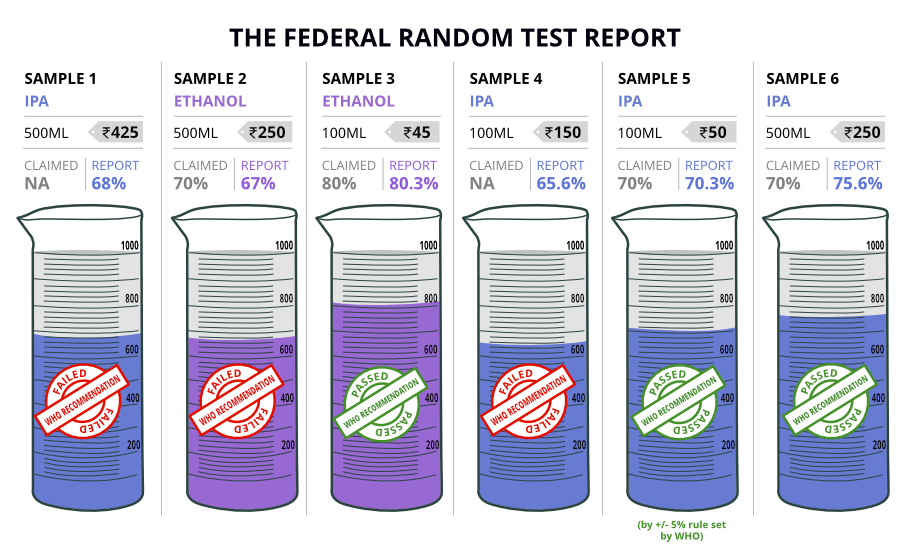
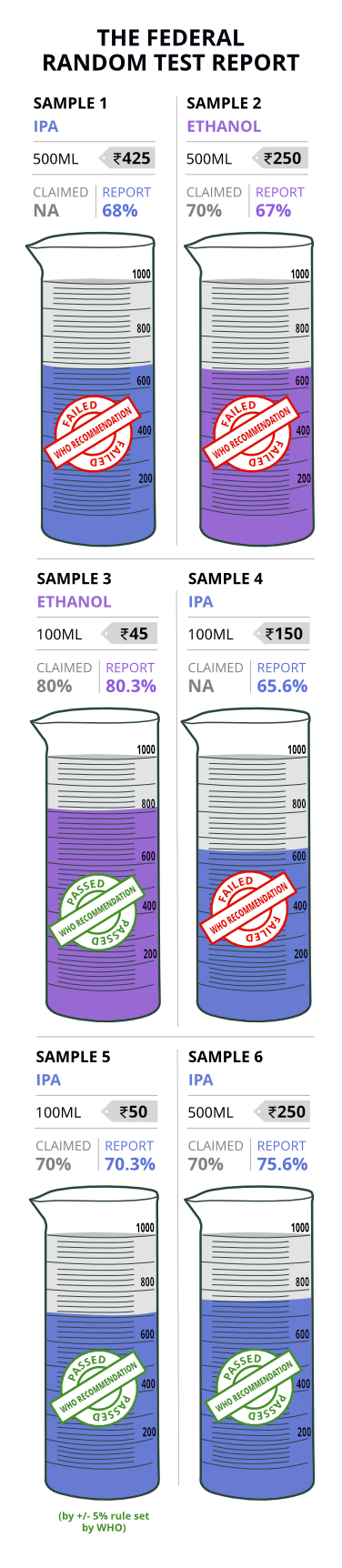
But, in the wake of the COVID-19 situation, the CDSCO (The Central Drugs Standard Control Organisation) has allowed manufacturers to use 24 other combinations using different chemicals. All states have to go by the CDSCO norms.
Missing manufacturing licence number
One major problem we noticed with the tested sanitisers is that, two out of the three brands which contained less than 70% alcohol, did not have the manufacturing license number in them. The third brand had the manufacturing license number, but we detected issues related to inspection of the product.
While the label mentioned that there was 70% alcohol content, our test results proved that there was only 67%. So, we spoke to the manufacturer for clarity. “We strictly adhere to the norms and we have 70% alcohol content in our product. Recently, there was even an inspection,” said Anbu (name changed), Production Manager of the company.
His claim of inspection being conducted got countered by the statement given by an official from the Tamil Nadu Directorate of Drugs Control. “Inspections have not been happening and will resume only after 30th of June because of the emergency situation. Generally, inspection is conducted both by the centre and the state, so we are unable to conduct them now. But, new licenses are being issued by the technical staff,” said Habeed Muhammed, Assistant Director of Drugs Control, Tamil Nadu Food Safety and Drug Administration Department.
He added that no one can sell sanitisers without getting a license issued and that all licensed manufacturers must mandatory have the manufacturing license number in the label.
With the COVID-19 situation increasing the demand for sanitisers, the government asked the country’s sugar industries and distilleries to prioritise supply of ethyl alcohol. Many of these companies have taken to manufacturing sanitisers with growing demand, but officials make it clear that they can sell sanitisers only after they acquire the license.
RELATED NEWS: Sanitiser over hunger: Centre to make ethanol from excess foodgrains
While the government is encouraging more players, they have also imposed a price ceiling of ₹112 for 200 ml bottles to make the product. Manufacturers say that the price ceiling is affecting them.
Naresh, manufacturer of a brand called Wellocare said, “We are unable to compete in the local market because of the price ceiling. Those manufacturing liquor have started manufacturing sanitisers too. They procure alcohol for a cheaper price than us. If alcohol costs ₹20-25 per litre for them, it costs ₹100 for us. When purchase price is different, they sell it at a lower price.” The manufacturers also say that price of nasal spray caps in bottles have increased because of the reduction in imports from countries like China.

Purity is very important
“It is possible that a lot of manufacturers take to diluting alcohol content because it is very costly. Good quality alcohol costs about ₹700 to ₹800 per litre, ” says Mathiarasu, senior principal scientist, CSIR Central Electrochemical Institute in Karaikudi
He adds, “Purity standard of alcohol is very important. According to WHO standards, the 99.8% purity of alcohol has to be maintained. Alcohol with 95% purity is also available, but the other 5% will have other organic molecules which are carcinogenic. If it comes in contact with the skin, people are prone to get carcinogenic diseases.”
Is it possible for a layman to identify good quality sanitiser?
No, says the scientist. Mathiarasu says, “One needs instrumentation facilities to check quality of the products. You need to rely on the company and go by the brand.” But, for people to trust the brands with licenses, it is very important that there is regular inspection, especially during COVID-19 times, when proper sanitisation can ‘Break the Chain’.


