
- Home
- News
- Analysis
- States
- Perspective
- Videos
- Education
- Entertainment
- Elections
- World Cup 2023
- Features
- Health
- Budget 2024-25
- Business
- Series
- NEET TANGLE
- Economy Series
- Earth Day
- Kashmir’s Frozen Turbulence
- India@75
- The legend of Ramjanmabhoomi
- Liberalisation@30
- How to tame a dragon
- Celebrating biodiversity
- Farm Matters
- 50 days of solitude
- Bringing Migrants Home
- Budget 2020
- Jharkhand Votes
- The Federal Investigates
- The Federal Impact
- Vanishing Sand
- Gandhi @ 150
- Andhra Today
- Field report
- Operation Gulmarg
- Pandemic @1 Mn in India
- The Federal Year-End
- The Zero Year
- Premium
- Science
- Brand studio
- Home
- NewsNews
- Analysis
- StatesStates
- PerspectivePerspective
- VideosVideos
- Entertainment
- ElectionsElections
- Sports
- Loading...
Sports - Features
- Budget 2024-25
- BusinessBusiness
- Premium
- Loading...
Premium
COVID-19 vaccine breakthrough infections—what we know and what we don’t
Vaccine breakthrough infections are infections in fully vaccinated individuals. For COVID-19, vaccine breakthrough infections are defined as infections occurring in individuals, after 14 days of the second dose of approved COVID-19 vaccine.

The COVID-19 pandemic and its impact globally has seen accelerated research and development of vaccines at a pace previously unheard of. This has resulted in a number of vaccine candidates being developed and approved by respective regulatory agencies in different countries for use on humans. Countries across the world have secured and authorised one or more of the many COVID-19 vaccines....
The COVID-19 pandemic and its impact globally has seen accelerated research and development of vaccines at a pace previously unheard of. This has resulted in a number of vaccine candidates being developed and approved by respective regulatory agencies in different countries for use on humans.
Countries across the world have secured and authorised one or more of the many COVID-19 vaccines. They could be either mRNA or DNA vaccines, adenovirus backbone vaccines or protein subunit vaccines. Active vaccination of global populations, starting with healthcare workers, commenced in many countries early this year. Early results suggest that the vaccines presently widely used are safe and effective in preventing severe disease and deaths.
India has also kept pace with the development of indigenous vaccines and also the scale of production of vaccines. In India, three vaccines have been approved for the public: AZD1222 /ChAdOx1) (Covishield), BBV152 (Covaxin), and Gam-COVID-Vac (Sputnik V), and recently, a DNA vaccine ZyDCoV-D.
It is heartening to note that daily vaccinations have touched over 1 crore doses recently and it is expected that a continued push to vaccinate a majority of the population, especially high-risk groups would go a great way in preventing deaths and hospitalisations.
Despite these accelerated pace of development and deployment, it is also notable that there is a stark inequality in access to vaccines across the world. As of September 4, about 40.3% of the world population have received at least one dose of vaccine and 27.6% have completed the schedule of two doses of vaccine.
As much as 5.34 billion doses have been administered globally, and 40.5 million doses are now administered each day. While high income countries are planning to initiate booster doses, only 1.8% of people in low-income countries have received at least one dose.
The continued evolution of SARS-COV-2 by accumulating genetic variants has given rise to many genetic variants of the virus. Genetic variants of the virus are part of the evolution of the virus and not all of the variants are of significance. Only a small subset of these variants associated with clinical and epidemiological relevance are considered as variants of concern (VoC).
The World Health Organisation has recently classified SARS-CoV-2 VoCs as Alpha (B.1.1.7), Beta (B.1.315), Gamma (P.1), and Delta (B.1.617.2). Broadly speaking, the variants of Concern or VoC are associated with much higher transmissibility and immune escape.
The emergence of delta (B.1.617.2) in India and globally and associated with a high transmissibility has given rise to a wave of infections in India and now also in many countries across the globe, resulting in a significant number of deaths. It is also observed that delta has dented the efficacy of vaccines as a number of cases of breakthrough infections have been reported.
What are vaccine breakthrough infections?
Vaccine breakthrough infections are infections in fully vaccinated individuals. For COVID-19, vaccine breakthrough infections are defined as infections occurring in individuals, after 14 days of the second dose of approved COVID-19 vaccine.
A breakthrough infection should not be a cause of panic, as it occurs in a minor fraction of vaccinated individuals. A significant large proportion of the vaccine breakthrough infections results in less severe disease, underscoring the fact that the approved vaccines are indeed effective in reducing infections, preventing serious disease and death due to COVID-19.
The biggest source of the prevalence of vaccine breakthrough infections have come from the data for Phase III clinical trials for vaccines. Data from the Phase III clinical trials for the vaccines currently being administered, (Covishield) suggest a breakthrough infection rate of 0.2% to 0.6 % and (Covaxin) presented a breakthrough rate of 0·77% in the cohort, post-vaccination.
BBV152/Covaxin vaccinees despite evidence of reduced neutralization, reports suggest that the vaccine confers protection against the Delta and Beta variant.
A study by Gajanan N Sapkal and team suggests that Covisheild/ChAdOx1 nCoV- 19 recipients who have pre-existing antibodies from a previous infection and/or cases of breakthrough infection after vaccination are protected against the delta variant.
NEW evidence on breakthrough infections indicates people who tested positive for #SARS-CoV-2 after 1 or 2 vaccine doses had significantly lower odds of severe disease, hospitalisation, or #LongCOVID than unvaccinated. https://t.co/MgHt7Ztqa0 @TheLancetInfDis pic.twitter.com/nt2b7jVrH9
— The Lancet (@TheLancet) September 1, 2021
The second source for prevalence comes from observational studies during the wave of infections in India and abroad. A recent preprint by Rajat Ujjainiya and team, which systematically followed up healthcare workers in a tertiary care centre in India during the recent wave of infections suggests that asymptomatic vaccine breakthrough infections were much more prevalent.
These findings from Delhi, India point out that being fully vaccinated does not confer absolute protection against asymptomatic infections. This also highlights that adherence to public health measures are still key to prevent the spread of the virus even in fully vaccinated individuals.
Why vaccine breakthrough infections occur
Multiple studies have underscored the safety and efficacy of COVID19 vaccines in preventing severe disease and death. While vaccines also considerably reduce the prevalence of infections, no vaccines have cent percent efficacy. Pre-existing antibodies from previous COVID-19 infections provide protection, yet vaccination confers additional protection by lessening chances of severe disease and death.
Recent observations have suggested that different variants of SARS-CoV-2 could have a differential effect on its ability to escape neutralising antibodies raised by vaccines or by previous infections and cause fresh infections.
Additional glitches in the human immune mechanisms are being explored in detail. Notwithstanding their occurrence, breakthrough infections typically come with mild to moderate symptoms. Breakthrough infections therefore do not undermine the efficacy of vaccines.
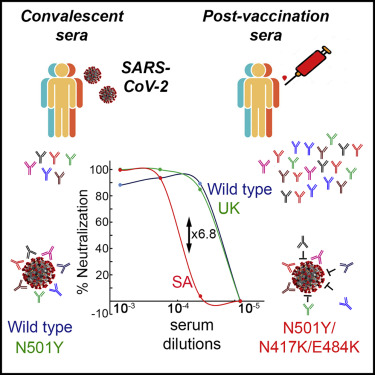
It is also imperative to understand that the vaccines are effective against current variants of concern like Delta which have been circulating across many countries worldwide.
In a study on vaccine efficacy against circulating variants like the delta variant by Manaf AlQahtani and team on the post vaccination breakthrough infections in Bahrain—which has authorised four vaccines (Astra-Zeneca (AZD1222 (ChAdOx1)/Covishield), Pfizer/BioNtech, Sinopharm/BBIBP-CorV and Sputnik V(Gam-COVID-Vac)—it concluded that from the cohort of vaccinated and unvaccinated participants, vaccinated individuals with breakthrough infections had significant reduction in disease burden, hospitalisation, ICU admissions and death noticeably prior to and after the dominance of the delta variant.
Vaccine hesitancy
It is disheartening to see that despite significant evidence to suggest that vaccines currently in use are safe and efficacious, certain sections of the population are still reluctant to get their shots.
With over 4.57 million deaths and 221 million cases of infections across the globe reported till September 6, 2021, vaccine hesitancy remains a much bigger problem than the pandemic itself.
In an article put together by Shingai Machingaidze, reluctance stems majorly from the miscommunication surrounding the possible side effects. Comparing studies from LMICs (Low and Middle Income countries) like Burkina Faso, Mozambique, Rwanda, Sierra Leone, Uganda India, Nepal, Nigeria, Pakistan and Colombia with that of USA and Russia (high income countries), it was found that vaccine acceptance in LMICs were 80.3%.
Pakistan and Burkina Faso both were the lowest in terms of vaccine acceptance (66.5%)..Racial and ethnic disparities in vaccination are not uncommon either. A study by Long H Nguyen and team revealed that owing to the vaccine hesitancy and access, COVID-19 vaccination was discernible amongst ethnic and racial minorities. When compared to the White, black participants are less likely to get the shot.
Covid-19 to continue
It is natural that newer variants of SARS-CoV-2 would emerge as the infections continue unabated across the globe, as a process of natural evolution of the pathogen.
While genomic surveillance can provide early insights into the emergence of such variants across the globe and can provide evidence to enable measures to contain the spread, the key to prevent the emergence lies with every one of us.
Strict and appropriate public health measures along with vaccination has been suggested as a significant approach to reduce the infections as well as significantly reduce severe disease and deaths associated with the infection.
The current pandemic also prompts us to acknowledge the fact that infectious diseases could lead to devastating consequences for the economy, livelihood and lives and no country can be seen in isolation in the globalised world. International cooperation to ensure vaccine equity should be one of the global priorities to end the pandemic.

