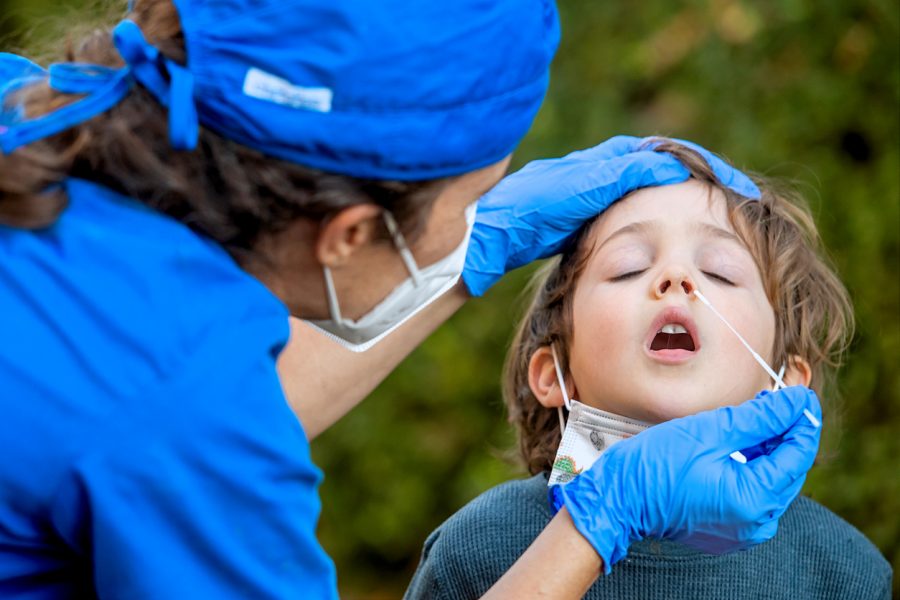
Inoculation for children to start soon; plan for booster dosage to be chalked out

COVID-19 vaccination for children and booster dosage for fully vaccinated adults are some of the major issues likely to discussed at a meeting of the government’s top advisory group on immunisation in the next two weeks, said a report.
Government sources also told the media that the vaccination for children with co-morbidities will commence first and will kick off from January, while other children will get inoculated from March.
The country’s drug regulator has already approved Zydus Cadila’s ZyCoV-D for those children who are 12 years and above, and has recommended granting emergency use authorisation (EUA) for Bharat Biotech’s Covaxin for children as young as two.
The vaccination campaign for children would have launched by now but the government was waiting for the Zydus Cadila’s vaccine to be delivered to them and for the Drugs Controller General of India’s (DCGI) approval for Covaxin (for children), said Dr N K Arora, chairperson, the National Technical Advisory Group on Immunization (NTAGI) to Mint. Over five COVID vaccines will also be available soon, which includes Biological E’s Corbevax and Novavax’s Covavax, he added. India, which is closely watching the progress of COVID vaccination among children in countries conducting the same, views children as a priority group since schools have opened in most states in the country.
Also read: Second dose overdue for more than 18 lakh people in Bengal
Meanwhile, the NTAGI is also set to chalk out a plan to provide additional doses of COVID-19 vaccine to adults. There has been a lot of discussions around the world on whether additional or booster dose is required.
On Monday, (November 22), ICMR Director General Dr Balram Bhargava however categorically stated that “there is no scientific evidence so far to support the need for a booster vaccine dose against COVID-19”. However, he underscored the importance of completing the second dose for India’s adult population. That is the priority of the government right now.
Earlier, he said at a meeting that both the doses are providing up to 98 per cent protection. “Our target is to get all those eligible vaccinated and till now about 80 per cent of the people have got their first dose,” he added. Union Health Minister Mansukh Mandaviya too recently echoed the same view. The minister said that adequate stocks were available and the aim was to complete the vaccination of the target population with two doses. After that, a decision on booster dose would be taken based on expert recommendation, he added.
Countries like the US, which had initially vetoed the booster shot, are however trying to broaden COVID-19 booster eligibility dose beyond those with comorbidities, the elderly and other high-risk groups. This is probably due to Pfizer’s study which showed that vaccine efficacy against symptomatic disease was restored to 95.6 percent when a booster was administered in a study involving 10,000 people over the age of 16. Also, there is the example of Israel, which reportedly fought back its Delta wave with a nationwide booster campaign.
The jury is still out on the subject since even as many scientists believe it’s the right time for the move; some feel the original vaccines are adequate enough to counter severe disease and death among the general population.


